Duchenne muscular dystrophy gene therapy approved by USFDA.
Yesterday it was a golden day in the medical profession and Bio pharmaceutical development Duchenne muscular dystrophy gene therapy was approved by USFDA. It has provided hope for life to billions of DMD affected children.
Untill 22-06-2023 there was no treatment available for Duchenne muscular dystrophy (DMD). on 22-Jun-2023 an innovative new medicine Gene therapy is approved by US FDA for Duchenne muscular dystrophy (DMD) in patients with confirmed X Chromosome gene is inactive.
What is Duchenne muscular dystrophy (DMD)
Duchenne muscular dystrophy (DMD) is a disease that takes a toll on the lives of younger children. The disease is diagnosed in the child at early age at 2 to 3 rd. year of age of a child, is diagnosed with Duchenne muscular dystrophy (DMD). Duchenne muscular dystrophy (DMD) is devastating on the family of the child whose parents suffer mental stress. As the age of the patient increases the disease gets prominent and the child at an early age starts losing muscular strength of his limbs. Most of the time requires help to mobilize from one place to another and depends on the parents to provide daily care, like bathing, wearing clothes, eating.
Disease progression of Duchenne muscular dystrophy (DMD).
As the age increases the muscles in the body, musculature of lungs, diaphragm, which are important in berating lose strength. The skeletal muscles are lost.
The death was certain for such children, without a treatment that is targeted to the root cause of the disease.
Duchenne muscular dystrophy (DMD) until yesterday had treatment with steroids like prednisolone and corticosteroid, to slow the muscle washing. And to mitigate the secondary symptoms those arise from loss of skeletal muscles.
What causes the disease Duchenne muscular dystrophy (DMD)?
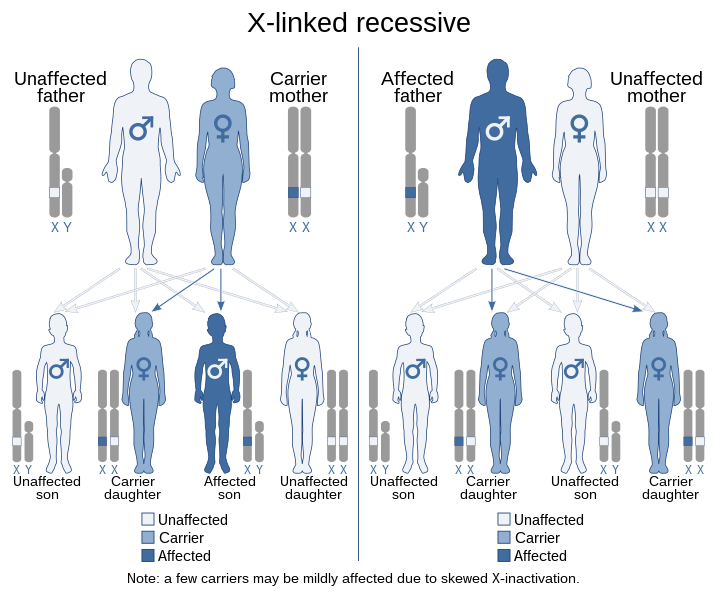
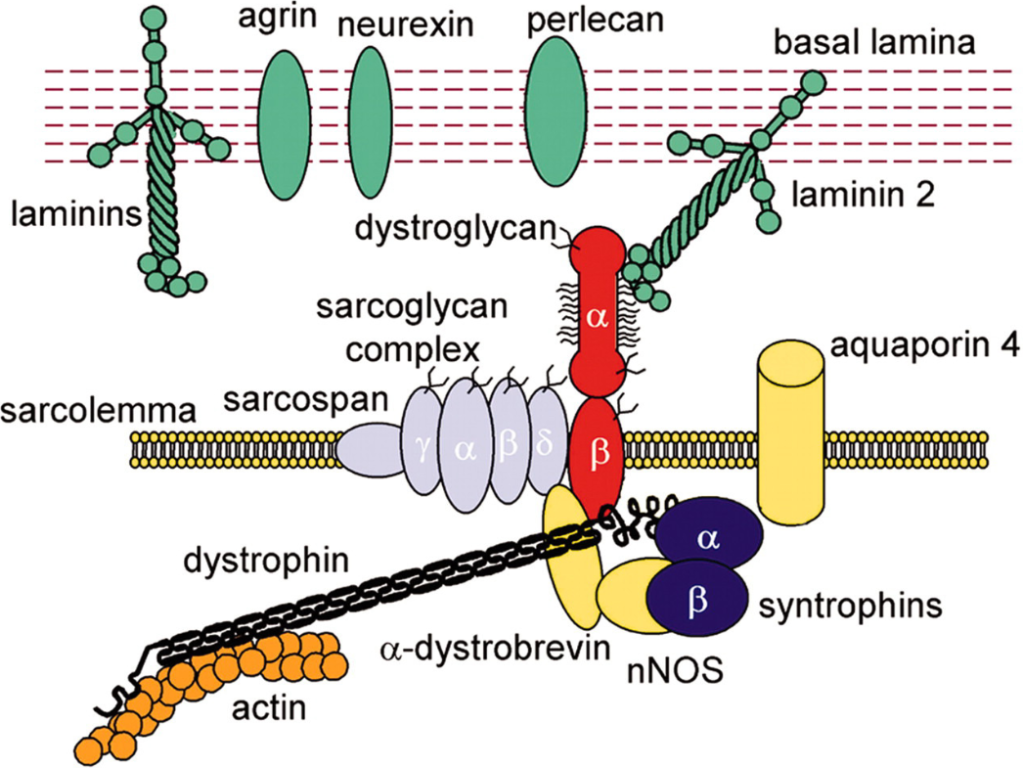
Patients affected with Duchenne muscular dystrophy (DMD) do not produce dystrophin, a protein fiber that binds muscles to bones and which imparts rigidity to muscles required for muscular contraction.
Without the dystrophin the skeletal muscles and smooth muscles lose their ability to perform their function of contraction and relaxation.
It is a genetic disorder, it is X linked, recessive (X chromosome is expressed in Male only while in female it is inactive completely or partly), that is if the chromosome X in one of the parent is deficient of the gene that produce the protein fibers dystrophin. Most of the time the male child is transferred the defective X chromosome either from carrier female or male. In rare conditions, female children do express the X chromosome but it is milder compared to male.
Gene therapy approved by US FDA for On 22-06-2023.
The US FDA has done great work by approving a gene therapy for treating the basal root cause of the Duchenne muscular dystrophy (DMD). FDA granted accelerated approval of Elevidys a Gene Therapy for Duchenne muscular dystrophy (DMD). The gene therapy product is developed by Sarepta Therapeutics, Inc. Gene is bio-synthesized by recombinant technology. The gene is inculcated into an adeno-associated virus vector which is not harmful, but infects the human muscle cells and it carries gene and delivers the gene in the muscle cells nucleus.
After which the gene is incorporated into human DNA. And starts producing dystrophin which is used by the patient’s body to build proper muscle structure and help in attachment and holding with bones.
There are few side effects of the drug but where there is no way for life there such inovative medicines must be developed.
This is second such gene therapy approved in past few days. Earlier gene therapy approved was for heamophillia
Referances: https://www.fda.gov/news-events/press-announcements/
Leave a Reply