-
Novel Drug siRNA that lowers hypercholesterolemia in patients with gene disorder heterozygous familial hypercholesterolemia (HeFH) or clinical atherosclerotic cardiovascular disease (ASCVD)
Novel drug for hypercholesterolemia which is a SiRNA is now approved by USFDA for heterozygous familial hypercholesterolemia associated with genetic disorder (inclisiran) injection of Novartis a silensing RNA inhibits the process of translation of protein so that faulty enzyme production stops which is responsible for lipid production and hyperlipidemia Basic constituent of the human body is…
-
First autoimmun antibody diagnostic test for type 1 diabetes.
ELISA Immunoassay test in detection of ZnT8Ab antibodies (zinc transporter 8 autoantibody) for diagnosis of Type 1 Diabetes. Type 1 diabetes is a form of diabetes which is one of very severe form of diabetes, wherein patients body do not produce insulin, or if at all it do is in very low amount. Insulin is…
-
Monoclonal Antibodies found to be reducing occurrence of migraine
There is very little treatment options available for migraine and often those are not so effective in all patients unlike like other disease treatment involves lot of change overs in medications. A protein called calcitonin gene–related peptide (CGRP)is responsible for migraine. Hence to prevent the occurrence of migrane monoclonal antibodies which target this peptide was…
-

Validation in Pharmaceutical Industry
Validation in Pharma Industry different types of validations regulatory requirements. Detail information on Validations in Pharma Validation in Pharma Industry is very important part of Pharmaceutical Quality Management System which in turn constitute quality assurance systems and intern Good manufacturing Practice guidelines. US FDA have given special emphasis on validation and it is prime requirement…
-
Validation in Pharmaceutical Industry Types of Pharma Validation
Pharmaceutical validation In pharmaceutical manufacturing industryPharma Validation regulatory requirements.Validation in Pharma Industry is very important part of Pharmaceutical Quality Management System which in turn constitute quality assurance systems and intern Good manufacturing Practice guidelines. US FDA have given special emphasis on validation and it is prim requirement of US FDA pre-qualification, as well as all…
-
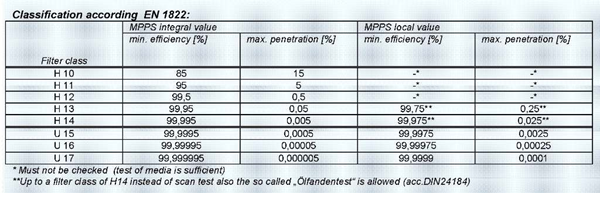
Pharmaceutical Area classification WHO guidelines
Pharmaceutical Area classification with respect to filters used as per WHO guidelines WHO gmp guidelines describes area in a pharmaceutical Manufacturing facility, as Level 1 level 2 and level 3 areas. Level 1 Area is place where there is no possibility of contamination of product, the product comes here after final packing if at all,…
-
Pharmaceutical Area classification with respect to filters used as per WHO guidelines
Pharmaceutical Area classification with respect to filters used as per WHO guidelines WHO gmp guidelines describes area in a pharmaceutical Manufacturing facility, as Level 1 level 2 and level 3 areas. Level 1 Area is a place where there is no possibility of contamination of product. Level 2 Area is an area where the raw…
-
New Options for HIV positive patients treatment will be available soon for daily dose of ART in HIV positive patients treatment
Treatment of HIV positive patients requires daily dose of ART (Antiretroviral Therapy) medicines which are a combination of many anti-retroviral drugs combined together. Combination ART drugs prescribed to HIV positive patients to keep their viral load under control and are very effective in providing protection against HIV to patients. Daily dose of antiretroviral medicines also has some…
-
Betalactam Antibiotics require separate manufacturing area in pharmaceutical manufacturing company
Why Betalactam Antibiotics require a separate manufacturing aria in a pharmaceutical manufacturing company?Chemistry behind the allergic reactions and mode of action of betalactum antibiotics: Answer:To avoid cross contamination of other products with betalactam antibiotics because A.The betalactum antibiotics may cause anaphylactic shock and anaphylaxis or may cause life threatening allergic reactions. B.The betalactum antibiotics molecules…
-
Automatic Insulin Pump that work in conjugation with blood glucose level sensor for children above 7 years age
Type 1 Diabetes patients are required to take Insulin through regular injections in hypodermic Insulin syringes since type 1 diabetes patients body do not produce or produce insufficient amount of Insulin hence blood glucose level remains higher, therefore type 1 diabetes patients are required to take dose of insulin through injections in daily repeated dosages.…